- Blog
- Metal slug 6 mame slow
- Fatin shidqia perahu kertas
- Vray sketchup 2016 beta
- Black and white game cheats
- Stardoll stardollar hack no download no survey
- Mortal kombat shaolin monks xbox
- The essential teachings of ramana maharshi
- Christmas jazz piano music with burning fire you tube
- Route a nier automata walkthrough
- Rufus taylor
- Boot usb iso thumb drive windows xp
- Legend of korra season 2 episode 1 full episode
- Hello neighbor mods
- Pathfinder society is the inner sea world guide legal
- Netflix ramayan
- Ucsra in codevision
- Girls with muscle maria villalba
- Native instruments komplete 10 high sierra
- Artistic marathi fonts
- Mobione studio ios emulator download
- Start easyweather data over
- How to calculate ppm ppb worksheet
- The dhandho investor espa-ol
- Mlb 2k12 pc roster update 2016
- Sumitra ramayan
- Moho pro 12 tutorials

This is the volume that results after the volume from the stock solution ( V 1) has been diluted with solvent or diluent to achieve a total diluted volume of the final solution ( V 2). Final Solution Volume (V 2) is the final volume of the diluted solution.Final Diluted Solution Concentration (C 2) is the concentration of the final diluted solution.Volume from Stock Solution (V 1) is the volume to be removed (i.e., aliquoted) from the concentrated stock solution.Stock Solution Concentration (C 1) is the concentration of the stock solution.Dilution factor may also be expressed as the ratio of the concentration of stock solution ( C 1) to the concentration of the final diluted solution ( C 2). It may be expressed as the ratio of the volume of the final diluted solution ( V 2) to the initial volume removed from the stock solution ( V 1), as shown in the equation above. Dilution Factor is the factor by which the stock solution is diluted.Jan 2008 - 38 What is the concentration of O 2(g), in parts per million, in a solution that contains 0.008 gram of O 2(g) dissolved in 1000.Dilution factor equation - ppb, ppm, ppt, pph grams of a solution having a concentration of 5 parts per million? Therefore, at least three cells must have values, and no more than one cell may be blank.

Enter appropriate values in all cells except the one you wish to calculate. Each calculator cell shown below corresponds to a term in the formula presented above. June 2009- 36 What is the total mass of solute in 1000. Dilution calculator - ppb, ppm, ppt, pph. June 2008- 38 What is the concentration of O 2(g), in parts per million, in a solution that contains 0.008 gram of O 2(g) dissolved in 1000. The reverse osmosis process has to overcome osmotic pressure to operate. June 2005 -42 What is the concentration of a solution, in parts per million, if 0.02 gram of Na 3PO 4 is dissolved in 1000 grams of water? grams of H 2O, what is the concentration of the resulting solution, in parts per million? If 0.025 gram of Pb(NO 3) 2 is dissolved in 100. Here was another one, but it was a multiple choice. It is technically wrong not to add the solute and solvents together to determine the mass of solution. Even though mathematically it didn't change a thing.
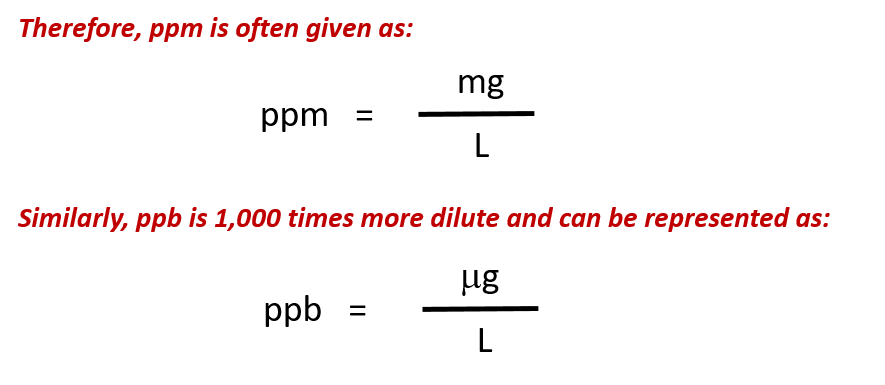
Calculate the molality of a solution prepared by dissolving 125 mL of pure methanol (density 0.791 g/mL) into 275 g of ethanol. Convert 2.4 ppm F-into molarity of fluoride ion. Īnswer-The equation above reads "grams of solution", you had to add the solute to the solvent in the denominator. Convert 0.0035 NaCl by mass into parts per million of NaCl. Your response must include both a correct numerical setup and the calculated result. In the space in your answer booklet, calculate the dissolved oxygen concentration of this solution in parts per million. An aqueous solution has 0.0070 gram of oxygen dissolved in 1000. Most students lost 1 of the 2 points on this one.Ħ6. Then June 2007, New York State threw this curveball. What is the concentration of a solution, in parts per million, if 0.02 gram of NaCl is dissolved in 1000. This is really just a plug and chug problem. Think about this 1ppm = 1 inch in 16 miles ppm parts per million ppb billion ppt trillion. This is used for very small concentrations of particles in solutions. Measuring Concentration: There are several different ways to measure and express the concentration. This is another way of determining concentration.